2]%2B%2B4.%2BTetrahedral.%2Bsp3.%2B[Cd(NH3)4]2%2B.jpg)
In molecules with more than three atoms, there are many more possible geometries. However, with a triatomic molecule (three atoms), there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule. sorry if this is confusing electro geo electronic geometry mol geo molecular geometry Learn with flashcards, games, and more for free. Molecular geometry and electronic geometry have different definitions. An example of the complexities which arise with polyatomic molecules is molecular geometry: how are the atoms in the molecule arranged with respect to one another? In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. The root anion for ClO 4 is the chlorate (ClO 3) ion. It comprises a chlorine atom which is bonded to four atoms of oxygen. 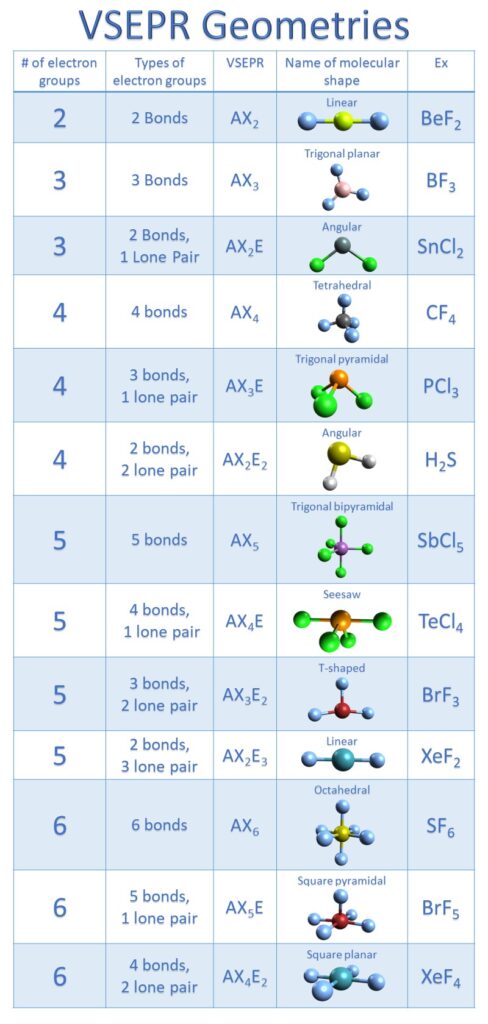
If there are no lone pairs and all orbitals are bonding, then the molecular geometry is the electronic geometry. ClO 4 is the chemical formula for the perchlorate ion. After calculating the electronic geometry from VESPR we can determine the molecular geometry based on the bonding orbitals. You also treat double and triple bonds as one group instead of 3 just like you did in molecular shape. However, with electron geometry (steric number) lone pairs and bonds are treated the same. A polyatomic molecule contains more than two atoms. Perchlorate ClO4- ion Lewis dot structure, molecular geometry or shape, electron geometry, bond angle, hybridization, formal charges, polar vs. In molecular shape (molecular geometry) you treat the electron’s lone pairs and the bonds as two different and separate things.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
